 
So, because the mass of hydrogen is close to 1 amu (atomic mass unit),1 gram of hydrogen contains about that same huge number of atoms. Hydrogen has an atomic mass of 1.01 atomic mass units. 
For every mole, there are 6.02*10 23 atoms, so in 1 gram of hydrogen there are 6.02*10 23 hydrogen atoms (in scientific notation) this is equal to 602000000000000000000000 hydrogen atoms. In kilograms, the molar mass of a hydrogen atom can be written as 0.001 kilograms per mole. The mass of an atom of hydrogen can also be expressed in molar mass units as one gram per mole. Multiply and round the answer to 5 significant figures.īecause the molecular mass of Hydrogen is 1 gram/mole, there is 1 mole of hydrogen in 1 gram of hydrogen atoms. In grams, the mass of an atom of hydrogen is expressed as 1.67 x 10-24. Multiply both sides of the equation by 1.0 g H to isolate the variable x on the right side.ġ.0000 g H * (6.022 x 10 23 atoms H / 1.0079 g H) = x atoms HĦ.0221 x 10 23 atoms H / 1.0079 = x atoms H Hydrogen atoms are also the smallest and lightest of all the atoms with only one electron. So, if there is 1.0079 grams of hydrogen per mole, then you can find out the number of hydrogen atoms in 1.0000 grams using the following proportion:Ħ.0221 x 10 23 atoms H / 1.0079 g H = x atoms H / 1.0000 g H By mass, hydrogen makes up about 75 of all matter in the Universe. 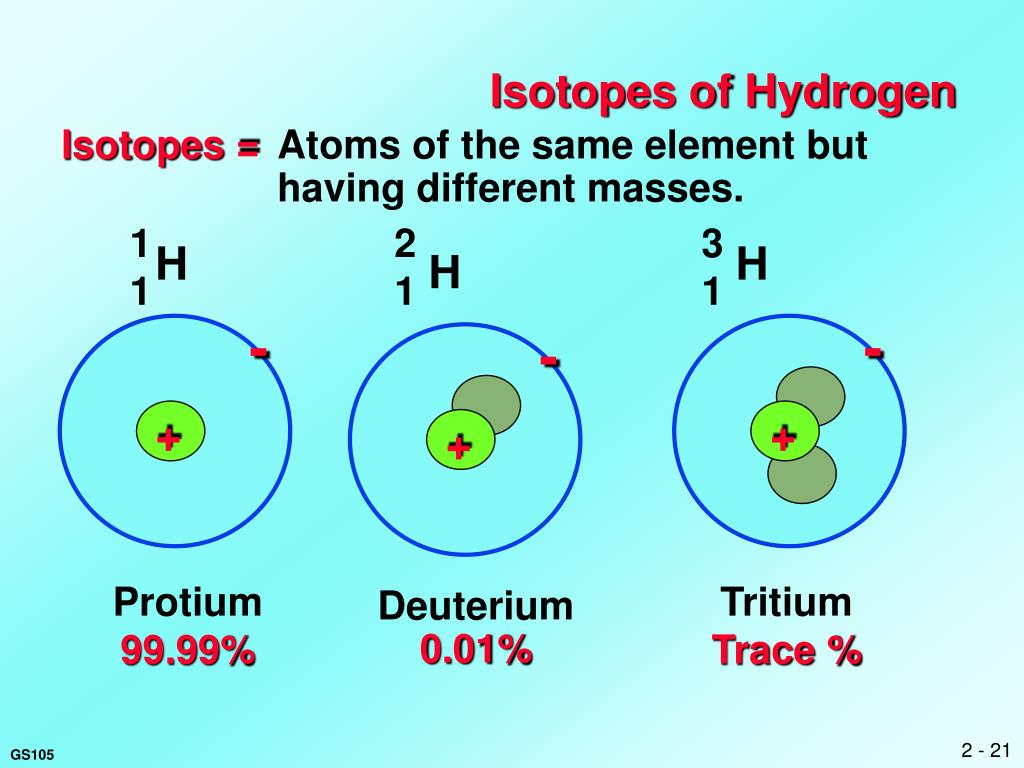
The atomic weight of hydrogen is 1.0079, which means that one mole of hydrogen atoms (or 6.0221 x 10 23 atoms of hydrogen) has a mass of 1.0079 grams. These isotopes with an additional neutron add a tiny amount to hydrogens average atomic weight. An even smaller proportion of hydrogen atoms are the isotope tritium, or hydrogen-3, which has one proton and two neutrons. Hydrogen has an atomic weight of 1.0079 rather than 1.0000 because a small proportion of hydrogen atoms are the isotope deuterium, or hydrogen-2, which has one proton and one neutron rather than just one proton. Using these values gives an atomic mass of 938.8 MeV/ $c^2$ for hydrogen, but the measured atomic mass of hydrogen is 939.0 MeV $/c^2$.How many hydrogen atoms would you find in 1g of hydrogens?Ī mole (Avogadros number) is the number that is equivalent to the number of atoms in 12 grams of pure carbon-12, which is always 6.022 x 10 23 atoms. 
Proton mass is 938.272 MeV/ $c^2$, electron mass is 511 keV/ $c^2$ and the ionization energy of hydrogen is 14 eV. The calculation does not add up, however. the ionization energy of hydrogen is featured. However, as there is only one nucleon in hydrogen, the contributions to B do not feature a any term from nuclear binding, only the binding energy of the single atomic state in hydrogen, i.e. If I take the hydrogen atom, and try to predict its mass this way, I find, unsurprisingly And where B is the net binding energy of a nucleus. Where subscripts $m_p$, e and n mean proton, electron and the neutron masses, respectively. of H + Atomic mass of S + Atomic mass of O Molar mass of H 2 SO 4 2 × 1. Let me elaborate: the atomic mass of some nucleus $^A _Z X_N$ is defined as Find important definitions, questions, meanings, examples, exercises and tests below for What is the mass of 0.5 mole of Hydrogen atom a)1.5 gb)0.5 gc)2.0 gd)1. Answer: One sulfuric (H2SO4) molecule has 2 hydrogen atoms, 1 sulfur atom. It appears that the mass of the simplest of the examples, hydrogen is not correctly produced, yet I cannot put my finger on what is the reason for this. After spinning around the atomic mass equation for calculating neutron separation energies, I have run into somewhat of a conundrum. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
